 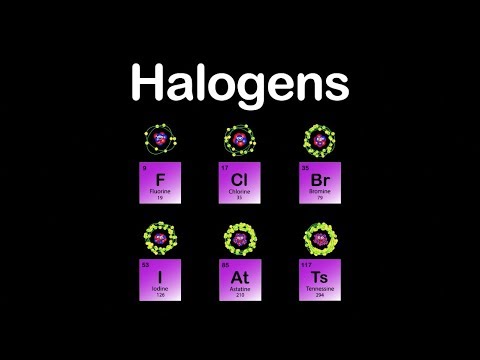
1: Derivation of the names of each of the halogens. The halogens / hldn, he -, - lo -, - dn / 1 2 3) are a group in the periodic table consisting of six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and the radioactive elements astatine (At) and tennessine (Ts), though some authors 4 would exclude tennessine as its chemistry is unknown and. 1 lists the derivation of the names of the halogens. The name alkali metals stems from an Arabic word. This is due to the formation of salts when they form compounds with a metal. These six elements can also be called Group 1 or Group 1A elements as well, because they are in the first vertical column on the periodic table. 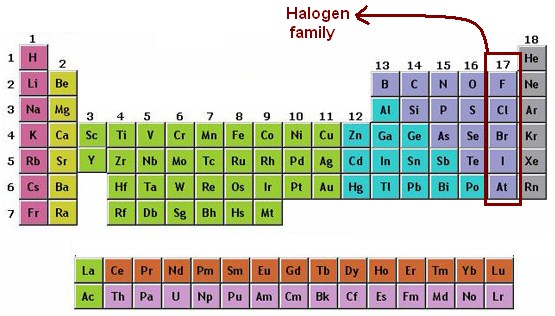
Other elements are not shiny, malleable, or ductile, and are poor conductors of heat and electricity. The Group 17 elements have a particular name: the halogens meaning born of salt. For example, many elements appear shiny, are malleable (able to be deformed without breaking) and ductile (can be drawn into wires), and conduct heat and electricity well. Many elements differ dramatically in their chemical and physical properties, but some elements are similar in their behaviors. (Francium, the last natural element to be discovered, was identified by Marguerite Perey in 1939.) When halogens react with metals, they produce a wide range of salts, including calcium fluoride, sodium. We will look at some of the physical and chemical properties of Halogens. Rhenium, one of their discoveries, was one of the last natural elements to be discovered and is the last stable element to be discovered. The word halogen means salt former or salt maker. The elements are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). They all contain five electrons in their outer p-subshell and commonly form. Thus, by investigating ores containing minerals of ruthenium (Ru), tungsten (W), osmium (Os), and so on, they were able to identify naturally occurring elements that helped complete the table. alkaline-earth metal, any of the six chemical elements that comprise Group 2 (IIa) of the periodic table. Halogens are a group of elements found in the periodic table. Their method was unique in that they did not only consider the properties of manganese, but also the elements horizontally adjacent to the missing elements 43 and 75 on the table. Elements in the periodic table are arranged by increasing atomic. German chemists Ida Tacke and Walter Noddack set out to find the elements, a quest being pursued by scientists around the world. The periodic table provides a visual means for displaying the elements found in nature or artificially produced in laboratories. Mendeleev had predicted, and others including Henry Moseley had later confirmed, that there should be elements below Manganese in Group 7. \): Elements in the periodic table are organized according to their properties.Įven after the periodic nature of elements and the table itself were widely accepted, gaps remained. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
